mechanical properties on food
In recent years, there has been a growing interest in the physical properties of food. A number of books and reviews dealing specifically with the subject have been published. Some physical properties of food such as viscosity in fluid flow, particle size in size reduction, thermal properties in heat transfer, diffusivity in mass transfer, etc.
Mechanical Properties on Food
Mechanical properties are the properties that determine the behavior of a food material when in contact with an external force. Mechanical properties can be used for processing (eg transport, size reduction) and consumption (texture, mouthfeel). The forces acting on the material are usually expressed as stress, i.e. the intensity of the force per unit area (N/m2 or Pa). Dimensions and units of stress are similar to pressure. Very often, but not always, the response of a material to stress is deformation expressed as strain. Strain is usually expressed as a dimensionless ratio, such as elongation as a percentage of the original length. The relationship between stress and strain is the subject of a science known as rheology.
Elastic Deformation
Elastic deformation appears instantaneously when there is pressure and disappears instantly by releasing the pressure. For some materials, strain is proportional to stress, at least for moderate deformation values. The elastic deformation is defined by Hooke’s law as:
E = Stress/Strain = (F/A0)/(∆L/L0)
E = Modulus Young , Pa
F = Force, N
A0 = Initial surface area, m2
ΔL = Length increase, m
L0 = Initial length, m
Plastic Deformation
Plastic deformation does not occur as long as the stress is below a limit value known as yield stress. Plastic deformation is permanent, that is, the workpiece does not return to its original size and shape when the stress is removed.
Viscous Deformation
Deformation (flow) that occurs instantaneously with pressure and is permanent. The strain rate is proportional to the stress.
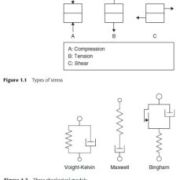
Stress types are classified according to the direction of the force in relation to the material. Normal stress is the stress that acts in a direction perpendicular to the surface of the material. Normal stresses are compressive forces if they act on the material and tensile forces if they act away from it. Shear stress acts in a parallel (tangential) direction to the surface of the material. The increase in deformation of a body under constant pressure is called creep. The decay of stress with time under constant strain is called relaxation.
Rheology Model
The stress-strain relationship in foodstuffs is usually complex. It is therefore useful to describe the actual rheological behavior of food with the help of simplifications such as drawing models. The models are constructed by connecting ideal elements (elastic, viscous, frictional, breaking, etc.) in series, parallel or a combination of both. Physical models are useful in the development of mathematical models (equations) for the description and prediction of the rheological behavior of complex foods.

Contributor: Daris Arsyada
Reference:
Berk, Zeki. 2008. Food Process Engineering and Technology. United States of America: Elsevier
water content in food
Water is the most abundant substance in most foods. Classification of food is divided into three groups according to their water content (high, medium and low humidity). Fruits, vegetables, juices, raw meat, fish and dairy fall into the high humidity category. Breads, hard cheeses, and sausages are examples of medium-humidity foods, while the low-moisture groups are dehydrated vegetables, whole grains, powdered milk and dry soup mixes.
The importance of the function of water in food goes far beyond the quantity in the composition of the food. Water is essential for the good texture and appearance of fruits and vegetables. In such products, loss of water usually results in lower quality. In other hand, water, becomes an important requirement for the occurrence and support of chemical reactions and microbial growth, and prevents chemical spoilage of food.
It is now well known that the effect of water on food stability cannot be related solely to quantitative water content. For example, honey containing 23% water is very stable in storage while dehydrated potatoes will undergo rapid decay at half-high water content. To explain the effect of water, parameters that reflect the quantity and ‘effectiveness’ of water are needed. This parameter is water activity.
Water activity aw is defined as the ratio of the vapor pressure of the food to the vapor pressure of pure water at the same temperature.
aw = P/P0
P = partial pressure of water vapor of food at temperature T
P0 = equilibrium vapor pressure of pure water at temperature T. The same type of ratio also determines the relative humidity of air, RH (usually expressed as a percentage):
RH = (P’/P0)*100%
P’ = Partial pressure of water vapor in the air
If food is in equilibrium with air, then p = p’ . Therefore the water activity of the food is equal to the relative humidity of the atmosphere in equilibrium with the food. For this reason, water activity is sometimes expressed as the equilibrium relative humidity, ERH.
aw = ERH/100
| Range aw | Product |
| above 0.95 | Fresh fruit, vegetables, milk, meat, fish |
| 0.90-0.95 | semi-hard cheese, anchovies, bread |
| 0.85-0.90 | hard cheese, sausage, butter |
| 0.80-0.85 | fruit juice, jelly, wet pet food |
| 0.70-0.80 | jam, dry cheese, legumes, plums |
| 0.50-0.70 | raisins, honey, seeds |
| 0.40-0.50 | almond nut |
| 0.20-0.40 | Non-fat milk powder |
| <0.2 | biscuits, roasted ground coffee, sugar |
Typical water activity in food
The main mechanisms that contribute to the decrease in water vapor pressure in food are solvent-solute interactions, binding of water molecules to the poles of polymeric substances (e.g. polysaccharides and proteins), adsorption of water on the solid surface matrix and capillary forces. In high-moisture foods, such as fruit juices, depression can be attributed entirely to water-solute interactions. If the food is considered an ‘ideal solution’, this water vapor pressure applies Raoult’s law:
P = XwP0
Where Xw is the water content (in mole fraction) of the food. Therefore the water activity of an ideal aqueous solution is equal to the molar concentration of water Xw. The water activity of foods with high humidity (with an aw of 0.9 or higher) can be calculated quite accurately by this method.
Contributor: Daris Arsyada
References:
Berk, Zeki. 2008. Food Process Engineering and Technology. United States of America: Elsevier